You probably see titanium every day without even realizing it. It's in your phone, your laptop, maybe even in the screws holding your glasses together. It's light, incredibly strong, and doesn't rust. Surgeons trust it enough to implant it within the human body. Fighter jets depend on it to stay airborne at supersonic speeds. Yet, here's the paradox: titanium is the ninth most abundant element on Earth. It's everywhere. So why is it so expensive? And why is making it so ridiculously difficult? The answer lies in the intricate chemistry and industrial challenges that have long hindered its mass production.
Key Takeaways
- Titanium is abundant but difficult to extract due to its reactivity with oxygen and other elements.
- The traditional Kroll process is energy-intensive, slow, and requires specialized equipment and controlled environments.
- Despite its remarkable properties, no efficient large-scale method has been developed to produce titanium at lower costs.
From Ore to Metal: The Complex Journey of Titanium
Titanium does not exist in nature as a pure metal. Instead, it is embedded within minerals, primarily in the form of ilmenite, a compound of titanium and oxygen. This mineral is typically found in sandy, dark deposits along beaches or ancient riverbeds. When extracted, it yields titanium dioxide, a white powder with applications ranging from paint and sunscreen to toothpaste. However, the transformation from this oxide to the metallic form is where the complexity begins.
For most metals, the extraction process is relatively straightforward: heat the ore in a furnace, allow impurities to burn off, and pour out liquid metal. Titanium, however, refuses to cooperate. When exposed to heat in the presence of oxygen, it does not purify; instead, it binds more tightly to the oxygen. Excessive heat can also cause titanium to absorb nitrogen and carbon from the air, resulting in a brittle, unusable material. This reactivity is the crux of the challenge in its production.
The Kroll Process: A Legacy of Innovation and Inefficiency
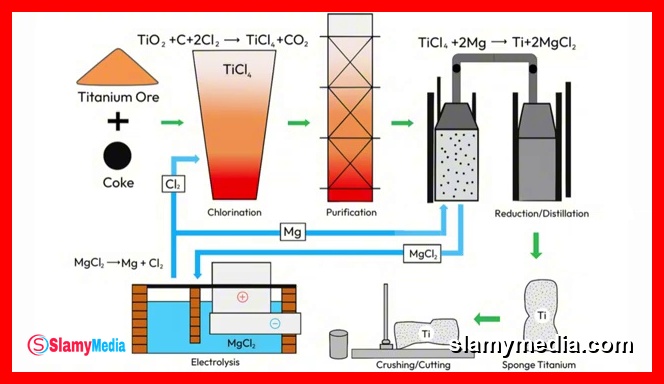
The breakthrough in titanium production came in 1940, when William J. Kroll developed a method that successfully extracted titanium from its oxide form. Known today as the Kroll process, it remains the primary method for producing titanium metal. Despite its historical significance, the process is notoriously inefficient. The Kroll process involves several stages, each requiring precise control and specialized conditions.
First, titanium dioxide is mixed with chlorine gas and coke (a form of carbon), and the mixture is heated to approximately 1,000 degrees Celsius. The chlorine reacts with the titanium dioxide, stripping away oxygen and forming titanium tetrachloride, a liquid that appears water-like but reacts violently with moisture in the air, producing thick white clouds. This compound is still not metallic and remains highly reactive.
Next, the liquid titanium tetrachloride is dripped into a sealed steel reactor filled with molten magnesium, heated to around 850 degrees Celsius. The magnesium acts as a stronger binder than titanium, and it effectively steals the chlorine away, leaving behind pure titanium and magnesium chloride as a byproduct. However, the resulting titanium does not emerge as a smooth ingot. Instead, it forms a spongy mass with numerous holes, mixed with leftover magnesium chloride. This resembles a giant metallic coral reef.
The purification process is far from complete. The sponge must be broken apart, crushed, and washed to remove the salts. It is then melted in a vacuum furnace to avoid contamination, yielding a solid block of usable titanium metal. The entire process takes days and is carried out in batches, not continuously. It demands extreme temperatures, toxic chemicals, and sealed environments. Each step must be meticulously controlled due to titanium's extreme reactivity with its surroundings.
The Economic and Industrial Implications of Titanium Production
This intricate and energy-intensive process is the reason titanium remains expensive. Its abundance does not translate to affordability due to the high cost of extraction and processing. In contrast, aluminum was once more valuable than gold in the 19th century. However, the development of an efficient extraction method using electricity in a continuous process led to a dramatic price drop, making aluminum ubiquitous. Titanium, despite its remarkable properties, has yet to see a similar breakthrough in production technology.
Scientists have explored numerous alternatives over the decades, seeking a more efficient method to produce titanium. Yet, the fundamental challenge of its reactivity persists. Until a breakthrough occurs, titanium will continue to be a rare and costly material, despite its prevalence in the Earth's crust. The journey from ore to metal remains a testament to the complexities of industrial chemistry and the enduring quest for materials that balance performance with economic viability.









Conversations (0)